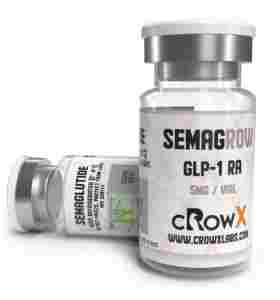
Tirzepatide 5 Peptide Plus USA

- Brand: Peptide Plus USA
Shipping Details
Your order will be delivered via USPS trackable service within 5-6 business days after payment processing.
More Information
| Product Features | |
| PACK | 5 mg 2 ml vial |
| SUBSTANCE | Tirzepatide |
Shipping
$35 flat rate
Payment Methods
Crypto, Venmo, Zelle, Credit Card, WU
Secure Shopping
Your data is protected with the highest security standards.
⚠️ WARNING: Health Risks of Anabolic Steroids
Anabolic steroids significantly impact the body and require professional supervision. Improper use may cause serious health risks including hormonal disruptions, liver stress, heart problems, and mental health changes. Always source steroids from reputable suppliers and use them responsibly if you choose to proceed.
-Important Note: The original text incorrectly identified Tirzepatide as a "glucagon receptor agonist." Tirzepatide is actually a GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptor agonist. Glucagon receptor agonism is a mechanism explored in other experimental peptides (like retatrutide), but not tirzepatide. I have corrected this factual inaccuracy in the revision below.
Tirzepatide 5 Peptide Plus USA
Disclaimer: This summary is intended exclusively for informational and scientific review purposes. Tirzepatide is an investigational compound in many laboratory contexts and must only be handled or administered under the strict supervision of licensed medical professionals. This text does not constitute medical advice.
Overview of the Compound
Tirzepatide represents a novel class of metabolic research agents known as dual "twincretin" receptor agonists. In research settings, the 5mg lyophilized vial is utilized to study advanced metabolic regulation. Unlike single-action agents, Tirzepatide is engineered to activate both the GIP and GLP-1 receptors natively found in the human body, creating a synergistic effect that researchers are evaluating for enhanced glycemic control and weight reduction.
Hypothesized Mechanisms of Action
Studies into Tirzepatide focus on how this dual-receptor activation optimizes natural metabolic and digestive responses through several pathways:
Synergistic Incretin Effect: By targeting both GIP and GLP-1 receptors, the compound heavily stimulates glucose-dependent insulin secretion from pancreatic beta cells while actively suppressing inappropriate glucagon release.
Gastric Emptying and Satiety: Tirzepatide delays gastric emptying, prolonging the physical sensation of fullness. Furthermore, receptor activation in the brain's satiety centers helps naturally reduce caloric intake, which is considered the primary driver of its weight-loss efficacy.
Insulin Sensitivity: Early research suggests that GIP activation, combined with significant weight loss, may directly improve overall insulin sensitivity in adipose tissue.
Primary Clinical Research Areas
| Research Area | Target Profile | Key Investigational Outcomes |
| Type 2 Diabetes | Subjects unresponsive to first-line treatments (e.g., Metformin). | Unprecedented reductions in HbA1c levels compared to single-agonist basal insulin or GLP-1 therapies. |
| Chronic Obesity | Subjects with or without T2D requiring significant weight intervention. | Substantial, dose-dependent reductions in total body weight and fat mass. |
| Metabolic Syndrome | Profiles exhibiting overlapping metabolic comorbidities. | Improvements in cardiovascular markers, including blood pressure and lipid profiles. |
Investigational Protocols (SURPASS & SURMOUNT Trials)
The clinical efficacy and safety profiles of Tirzepatide are largely defined by the extensive SURPASS (diabetes focus) and SURMOUNT (weight management focus) clinical trial programs.
Administration: Protocols standardly utilize a once-weekly subcutaneous injection, which has been shown to yield high compliance rates in long-term studies.
Dose Escalation: Research protocols mandate a slow titration schedule, typically starting at a low induction dose and increasing incrementally every four weeks to mitigate initial gastrointestinal distress.
Safety Profile and Contraindications
Due to its potency, the compound is researched under strict medical monitoring with specific attention to the following risks:
Gastrointestinal (Common): Nausea, diarrhea, vomiting, and decreased appetite are frequently observed, particularly during the dose-escalation phases. These generally subside as the body acclimatizes.
Systemic Risks (Rare): Serious investigational warnings include potential risks for acute pancreatitis, gallbladder disease, and renal impairment secondary to severe dehydration.
Contraindications: Tirzepatide is strictly contraindicated in subjects with a personal or familial history of Medullary Thyroid Carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Research Quality Standards
For accurate and reproducible laboratory results, researchers mandate strict purity and handling standards for investigational compounds:
Purity Verification: High-Performance Liquid Chromatography (HPLC) testing is required to verify concentration and ensure structural integrity without impurities.
Stability: The compound is typically supplied as a lyophilized (freeze-dried) powder in a sterile vial, requiring careful reconstitution with bacteriostatic water prior to application.
Conclusion
Tirzepatide stands at the forefront of modern metabolic research. By leveraging the synergistic effects of both GIP and GLP-1 pathways, it has demonstrated the potential to outperform traditional single-action therapies in both glycemic control and weight management, marking it as a critical subject of ongoing endocrine and metabolic study.